
Harvard’s new AI model identifies potential treatments for rare diseases using existing drugs
Recent advancements in biological AI models have shown significant potential in the development of new medications. But can these technologies also be leveraged to find treatments for rare diseases using drugs that are already available? Researchers at Harvard Medical School aimed to explore this possibility with their new foundation model, TxGNN, which is specifically designed to identify existing drugs that could treat rare diseases lacking current therapies.
The TxGNN model, introduced in a paper published in Nature, is considered the first AI system to focus on repurposing already approved drugs to target rare diseases for which there are no treatments presently available on the market.
Although each rare disease individually affects a small number of people, the collective impact of the over 7,000 classified rare conditions is significant, affecting around 300 million people worldwide, according to The Lancet. Yet, only 5–7% of these conditions have drugs that are currently approved by the Food and Drug Administration (FDA). Interestingly, nearly one-third of all drugs approved by the FDA eventually gain approval for multiple uses, with some being applied to as many as 10 different indications, the authors of the study explain.
However, the challenge lies in the fact that discovering new applications for these drugs has historically been an unpredictable and serendipitous process. As the research team describes, it often relies on healthcare professionals accidentally uncovering these new uses while treating people.
“Predicting the efficacy of all drugs against all diseases would enable us to select medications with fewer side effects, design more effective treatments that target multiple points within a disease’s pathway, and systematically repurpose existing drugs for new therapeutic purposes,” the authors note.
The rise of AI-driven breakthroughs, such as Google’s AlphaFold protein prediction model, which won the Nobel Prize for Chemistry this year, has ignited an explosion of generative AI applications in drug discovery. Numerous companies are also developing models aimed at drug repurposing, according to GlobalData.
The Harvard team behind TxGNN claims their model is approximately 50% more effective than existing repurposing models in identifying potential drug candidates. Additionally, it is reported to be 35% better at predicting contraindications, which are reasons a specific drug may not be appropriate for an individual.
What sets TxGNN apart is its broader scope compared to many other AI models designed for drug discovery. As explained in a Harvard announcement of the research, while most models tend to concentrate on a single disease or a small group of related conditions, TxGNN is designed to comprehensively identify shared “mechanisms based on genomic underpinnings” between rare diseases and more common, better-understood conditions.
The tool has been trained on publicly available data sources such as DNA information and clinical notes and was validated using nearly 1.3 million de-identified patient records from Mount Sinai Hospital in New York. A notable feature of the TxGNN model is its explainer component, which clarifies the reasoning behind each prediction, offering a step-by-step breakdown of the decision-making process.
In a bid to encourage further scientific discoveries, the team has made TxGNN freely available to other researchers.
“This is precisely where we see the promise of AI in reducing the global disease burden, in finding new uses for existing drugs, which is also a faster and more cost-effective way to develop therapies than designing new drugs from scratch,” said Marinka Zitnik, one of the paper’s authors and an assistant professor of biomedical informatics at Harvard Medical School’s Blavatnik Institute.
This initiative highlights the potential of AI not just in the development of entirely new drugs but also in identifying existing medications that could be repurposed, offering faster, more efficient pathways to treatment for people living with rare diseases.
Read More
Surge in digital health investment as purchasers focus on value, outcomes, and patient-centred care
The latest survey from the Peterson Health Technology Institute (PHTI) highlights a significant increase in the planned investment in digital health solutions by health plans, employers, and healthcare systems. This upward trend is expected to continue throughout the coming year as these groups prioritise value and measurable outcomes in their purchasing decisions.
Detailed Insights from the 2024 State of Digital Health Purchasing Survey
The 2024 State of Digital Health Purchasing survey, developed in collaboration with NORC at the University of Chicago, offers a comprehensive analysis of current trends in digital health adoption. The survey not only sheds light on the evolving landscape of digital health but also provides key data on spending, contracting approaches, and future priorities in this rapidly growing field.
Ongoing Expansion in Digital Health Spending
This survey gathered responses from 332 decision-makers responsible for acquiring digital health solutions. The findings reveal a continued commitment to digital health innovation and investment across a broad spectrum of purchaser groups. Notably, the data indicates:
- Employers: An overwhelming 97% of employers plan to maintain or increase their digital health expenditure.
- Health Systems: A significant 86% of healthcare systems intend to sustain or grow their digital health spending.
- Health Plans: 84% of health plans also expect to maintain or expand their investment in these technologies.
The broad adoption of digital health solutions reflects the increasing recognition of their role in enhancing patient care and improving operational efficiencies across the healthcare sector.
Factors Fueling the Surge in Digital Health Investment
Several key drivers underpin this surge in investment:
- Rising Consumer Demand: The growing appetite among individuals for accessible and convenient digital health solutions is a major catalyst for increased spending across all purchaser groups.
- Improving Health Outcomes: Digital health technologies are widely acknowledged for their potential to elevate the quality of care and improve health outcomes for people, which is a strong motivator for purchasers.
- Economic Efficiency: Particularly for health plans, the ability to reduce costs through the use of digital health solutions is a primary reason for heightened investment.
These factors highlight the dual appeal of digital health solutions: not only do they meet the needs and preferences of individuals, but they also promise better outcomes and cost savings for healthcare providers and payers.
Trends in Contracting and Performance Measurement
The survey also reveals emerging trends in how digital health contracts are structured and how their performance is assessed:
- Short-Term Contracts: The majority of contracts between purchasers and digital health providers have a duration of two years or less. This allows purchasers to regularly evaluate the effectiveness of the solutions and adjust their strategies as needed.
- Risk-Based Contracts: Performance-based contracts, which link payment to the achievement of specific outcomes, are increasingly common. The survey indicates that 79% of purchasers are using these risk-based arrangements for at least some of their digital health solutions.
- Outcome-Focused Evaluations: Health plans and healthcare systems are particularly focused on health outcomes when evaluating the performance of their digital health contracts. In contrast, employers are more likely to assess success based on user adoption rates and engagement levels.
This emphasis on performance and outcomes is part of a broader trend towards evidence-based decision-making in healthcare, ensuring that digital health solutions deliver tangible benefits to both individuals and organisations.
Top Priorities in Disease Areas
The survey also highlights the priority areas where digital health solutions are being deployed. Purchasers are focusing on the following key conditions and areas of care:
- Diabetes
- Preventive Care
- Mental Health
- Cardiovascular Disease
- Primary Care
These areas reflect the most pressing healthcare needs that digital health solutions are being leveraged to address, offering potential for both improved health outcomes and enhanced healthcare delivery.
Expert Perspective: Bridging Demand and Evidence-Based Solutions
Commenting on the findings, Caroline Pearson, Executive Director of the Peterson Health Technology Institute, emphasised the opportunity for digital health companies to align more closely with the needs of purchasers. She stated:
“Health plans, employers, and providers want to meet continued demand for digital health solutions, while ensuring the solutions they purchase have strong evidence that they improve health and generate economic efficiencies. This survey shows that there is a valuable opportunity for digital health companies to better align their offerings with what purchasers need and want, which in turn, will allow more people to access these tools and the benefits they can bring.”
Pearson’s remarks underline the critical importance of providing digital health solutions that are not only innovative but also backed by robust evidence of their effectiveness in improving health and creating efficiencies in care delivery.
Conclusion: A Growing Market with Clear Priorities
The 2024 State of Digital Health Purchasing survey provides a detailed picture of a rapidly expanding market, where purchasers are increasingly focused on value, outcomes, and patient-centred care. As digital health solutions continue to evolve, those that can demonstrate their ability to enhance health outcomes, engage users effectively, and deliver economic benefits will be in high demand. For healthcare systems, employers, and health plans alike, the message is clear: investment in digital health is here to stay, and the focus on performance and measurable outcomes will only grow stronger.
Read More
Digital pathology to enhance precision and efficiency in diagnostics
NYU Langone Health has revolutionised disease diagnosis through the launch of its digital pathology programme, moving from traditional microscope-based examination to high-definition digital imaging. This transformation allows for real-time sharing of diagnostic images across the hospital network, bringing unprecedented clarity in the viewing of human tissue samples, while also reducing diagnosis time and fostering collaboration. This development enhances a critical yet often overlooked aspect of patient care.
“For as long as pathology has existed, we’ve been bent over microscopes in our offices, manually reviewing slides that have been physically delivered to us,” said Dr. Syed T. Hoda, Director of Digital Pathology, Clinical and Bone and Soft Tissue Pathology at NYU Langone. Dr. Hoda is one of about 100 bone and soft tissue pathologists in the United States. “As of this week, images can be sent to the appropriate pathologist in real-time, regardless of where a patient is being treated in our network.”
This digital shift is especially timely, as the healthcare system is grappling with a shortage of pathologists amidst rising disease rates. A limited number of specialists are tasked with confirming diagnoses that impact millions of people. The adoption of digital pathology not only boosts the efficiency of pathologists’ workloads, but it also addresses risks associated with physical slides, such as storage complications, degradation over time, and delays in delivering results. These improvements will contribute to a more resilient healthcare system.
In addition to enhancing efficiency, the programme is also creating new employment opportunities. NYU Langone is hiring 24 digital pathology coordinators, introducing a new career path within the healthcare industry.
Dr. Joan F. Cangiarella, Vice Chair of Clinical Operations in the Department of Pathology at NYU Grossman School of Medicine, highlighted the intense nature of pathologists’ work. “Our team examines tissue samples from individuals we may never meet, agonising over microscopic details that could have life-altering consequences. Now, with digital pathology, we can perform these tasks at a much higher level, further enhancing the exceptional care we already provide.”
This transition to digital also paves the way for artificial intelligence (AI) to assist in diagnostics, offering an exciting prospect for future advancements in healthcare.
“As one of the first health systems in the United States to fully adopt digital pathology, NYU Langone is advancing disease detection for its patients and, through the integration of AI, initiating a paradigm shift that could help alleviate an impending bottleneck in the healthcare system,” said Nader Mherabi, Executive Vice President and Vice Dean, Chief Digital and Information Officer at NYU Langone.
Exploring the potential of AI, Dr. Sean Hacking, Director of Digital Pathology Research, elaborated: “This programme will expand our diagnostic toolkit. Digital images will fuel the AI models we’re developing to detect cancer and other diseases at the earliest, most crucial stages, which can profoundly affect the treatment options available to individuals.”
The digital pathology programme aims to reduce the time between testing and treatment, improve collaboration among medical teams, and potentially increase the direct involvement of pathologists in patient care. Already, the impact is being felt across the hospital network.
“Just yesterday, I attended our first fully ‘digital’ tumour board,” shared Dr. Hoda. “I was able to instantly access entire cases while we discussed patients in real-time. In the past, retrieving glass slides from storage could delay clinical decisions by hours or even days. For someone awaiting a diagnosis, that wait can feel like an eternity.”
NYU Langone Health has established a long-term partnership with Philips to integrate cutting-edge digital pathology technology. Martijn Hartjes, Clinical Informatics Business Leader at Philips, echoed Mherabi’s enthusiasm, describing digital pathology as a foundation for expanding the capabilities of pathologists.
“NYU Langone Health is setting a new standard for precision diagnosis and treatment by fully embracing digital workflows in pathology. In other laboratories, we’ve seen efficiency improvements of 15 to 25 percent per case,” Hartjes said. “Digital pathology also supports the integration of AI, which can further enhance efficiency and unlock new possibilities in disease detection. At Philips, we understand the challenges of transitioning from analogue to digital processes, so we are committed to delivering technology and services that simplify this transition and empower clinicians to provide better care to more patients.”
NYU Langone Health is a leading, patient-centred academic medical institution, recognised for its excellence in patient care, education, and research. For three consecutive years, Vizient, Inc. has ranked NYU Langone as the No. 1 comprehensive academic medical centre in the United States. U.S. News & World Report has also placed nine of its clinical specialties among the top five nationwide. The health system offers a wide range of medical services across six inpatient locations, the Perlmutter Cancer Center, and more than 300 outpatient facilities across the New York area and Florida. NYU Langone’s network also includes two medical schools and a vast research enterprise.
Read More
New Regulatory Innovation Office to accelerate AI and emerging technologies in UK healthcare
The UK Government, led by Science and Technology Secretary Peter Kyle, has announced the establishment of a new Regulatory Innovation Office (RIO) aimed at facilitating the safe deployment of artificial intelligence (AI) and other advanced technologies within the healthcare sector. This new initiative is designed to streamline the regulatory process, allowing innovative products and services to reach the market more quickly while ensuring that safety and efficacy standards are maintained.
The RIO is set to play a key role in reducing bureaucratic barriers for businesses looking to introduce new technologies, thus enabling faster access to these advancements for the public and contributing to economic growth. The Department for Science, Innovation and Technology (DSIT) issued a press release on 8 October 2024, stating that the RIO will “support regulators to update regulation, speeding up approvals, and ensuring different regulatory bodies work together smoothly.”
The RIO’s initial focus will encompass four key areas of technology: AI and digital innovations in healthcare, connected and autonomous technology, engineering biology, and space. The office will collaborate closely with the Department of Health and Social Care to ensure that AI solutions, such as advanced AI training software for surgeons, are implemented safely within the healthcare system.
Peter Kyle emphasised the broader impact of RIO’s work, stating: “From breakthroughs that could help doctors diagnose illnesses earlier to satellite navigation for more accurate weather forecasting and getting emergency supplies to where they are needed, quickly and effectively, RIO will make sure UK companies are at the forefront of the next generation of technologies.” His statement highlights the potential for AI and emerging technologies to revolutionise various sectors, including healthcare, logistics, and emergency response.
The DSIT is currently seeking applications for the RIO’s first chair. This individual will be tasked with “leading the charge in backing business and safe innovation, and working with regulators and partners to shape a regulatory environment that is fit for the future.” This leadership role will be crucial in guiding the RIO’s efforts to create a regulatory framework that both supports innovation and ensures safety for users.
The new office aims to facilitate better coordination among different regulatory bodies and eliminate outdated regulations that may hinder innovation. The RIO will provide continuous feedback to the government on regulatory barriers that need addressing and set priorities for regulators to align with the government’s broader objectives. Additionally, it will support regulators in developing the necessary skills and capabilities to manage emerging technologies, thereby fostering economic growth.
The RIO will be housed within DSIT, integrating the existing roles of the Regulatory Horizons Council and the Regulators’ Pioneer Fund. By consolidating these functions, the RIO aims to create a more cohesive approach to regulating new technologies.
On the same day as the RIO’s announcement, the government also detailed its approach to regulating quantum technologies, in response to a report from the Regulatory Horizons Council on this field. This represents an initial step towards regulating innovations in quantum technology, which is expected to play a pivotal role in developing powerful computational capabilities across sectors, from healthcare to national security.
In addition to the RIO, the UK government has been actively investing in the advancement of quantum technology. In July 2024, DSIT announced a £100 million funding initiative to support research hubs dedicated to quantum technology. These hubs will focus on applications like quantum-enhanced blood tests, faster MRI scanning technology, and advanced surgical procedures and treatments. Such innovations promise to bring significant improvements to the healthcare sector, enabling more accurate diagnostics and novel treatment methods.
In parallel, the Medicines and Healthcare products Regulatory Agency (MHRA) has also taken steps to adapt its regulatory approach to emerging technologies. In May 2024, the MHRA launched the “AI Airlock,” a regulatory sandbox designed to address the unique challenges associated with medical devices that use AI. This initiative is intended to create a controlled environment where developers and regulators can collaborate to ensure that AI-driven medical devices meet safety and performance standards before they reach the market.
With the RIO and initiatives like the AI Airlock, the UK is positioning itself as a leader in the safe adoption of AI and other cutting-edge technologies in healthcare. By fostering a more adaptable and forward-thinking regulatory environment, the RIO aims to ensure that the benefits of these innovations are realised more swiftly, improving patient care and outcomes while supporting the growth of the UK’s technology sector.
Read More
Proposed staged rollout of Eli Lilly’s Mounjaro by NHS England includes digital solutions for wider access
The National Health Service (NHS) in England is exploring a phased introduction of the obesity treatment tirzepatide (Mounjaro, developed by Eli Lilly), driven by concerns over potentially high demand for the medication. The phased approach aims to ensure that individuals with the greatest clinical need receive the treatment first, while also preparing the system to meet the broader demand effectively.
The National Institute for Health and Care Excellence (NICE) initiated a three-week consultation on these proposals from NHS England, with final guidance expected to be published in December, 2024. This consultation period is intended to gather input from stakeholders on how best to manage the introduction of tirzepatide within the NHS framework.
Clinical Effectiveness and Pricing
Tirzepatide has shown greater effectiveness in promoting weight loss compared to diet and exercise support alone, as well as when compared with semaglutide alongside similar lifestyle support. Clinical trials have highlighted its potential to significantly aid people with obesity in achieving weight reduction. In its draft guidance issued in June, an independent committee from NICE recommended the use of the weekly injection for individuals with obesity. The treatment is priced at £122 per month for the maximum dose of 15 mg.
Eligibility Criteria for the Rollout
In its application to NICE, NHS England has outlined a staged approach for the rollout of tirzepatide. Initially, the drug will be made available to individuals with a body mass index (BMI) over 40 who also have at least three out of four specified weight-related health conditions: hypertension, dyslipidaemia, obstructive sleep apnoea, and cardiovascular disease. In subsequent stages, eligibility will be expanded to include those with a BMI over 40 plus two weight-related conditions, and eventually those with a BMI over 40 plus one weight-related condition.
This prioritisation aims to ensure that those with the most pressing health needs are given access to the medication first. NHS England estimates that nearly 250,000 people could be eligible for tirzepatide during the first three years of its phased introduction.
Exploring Digital and Community Services for Broader Access
To facilitate a smooth rollout and ensure timely delivery of the medication, NHS England is exploring innovative ways of administering tirzepatide, including digital and community-based services. These new models of care will be tested and evaluated to determine the most suitable and cost-effective approaches for providing weight loss support to an increasing number of individuals over the next nine years. The aim is to ensure that those eligible for the treatment receive the necessary dietary and exercise support alongside the medication, whether through primary or secondary care services.
NICE has acknowledged that the proposed length of the rollout is “unprecedented.” The extended timeframe is intended to ensure that resources are effectively managed and that the NHS can adapt its service delivery to accommodate the large number of people who may benefit from the new medication.
Reactions from Health Experts and Stakeholders
Sam Roberts, Chief Executive of NICE, emphasised the potential of this new generation of weight loss treatments to bring significant health benefits to people with obesity and to prevent the development of more serious health complications. Roberts stated, “Because of the very large number of people who could potentially benefit, NICE accepts that a phased rollout is required. However, we now need to hear from stakeholders on the proposals we have received from NHS England.”
Stephen Powis, NHS National Medical Director, supported the phased approach, highlighting its focus on those with the highest clinical need. He noted that this method allows time for the NHS to develop “new and innovative services through which other weight loss treatments can also be delivered.” These innovations aim to streamline the integration of tirzepatide into existing healthcare services and to accommodate future advances in weight management.
Simon Cork, a senior lecturer in physiology at Anglia Ruskin University, described the planned rollout of tirzepatide as a “key milestone in the future of obesity treatment.” He stressed the importance of providing comprehensive support to those receiving the medication, including tailored exercise programmes and personalised dietary guidance. Cork warned, “Without this high level of wraparound care, the NHS risks wasting money on these medications by not achieving the highest levels of weight loss possible. It is not clear whether the NHS has the capacity to provide this high level of wraparound care in the community to achieve this.”
A Strategic Move for Long-term Impact
The proposed phased rollout of tirzepatide represents a strategic move to address obesity care within the NHS while balancing demand and resource availability. By focusing initially on individuals with the highest need and considering the use of digital platforms to enhance accessibility, the NHS aims to maximise the impact of tirzepatide on public health. The feedback gathered during the NICE consultation will play a critical role in shaping the final approach, ensuring that the introduction of tirzepatide aligns with the needs of both patients and the healthcare system.
As the rollout progresses, the NHS and NICE will continue to evaluate and adjust their strategies, seeking to provide effective obesity care to a wider range of individuals while maintaining a sustainable approach to managing this new and promising medication.
Read More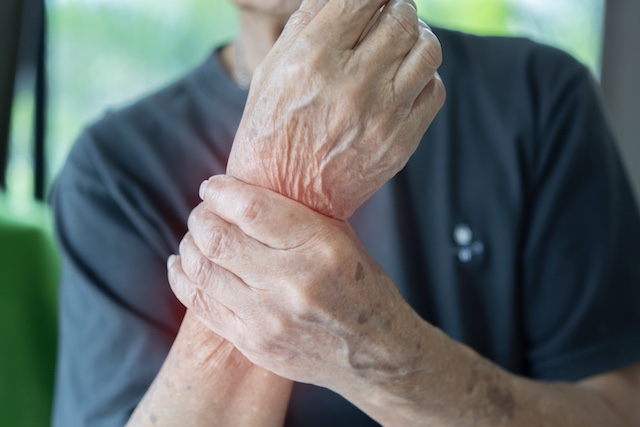
£1.2 million grant fuels development of AI platform for early arthritis detection
Henley Business School has secured a significant grant of £1.2 million to develop an innovative machine learning system aimed at improving the early detection and referral of rheumatic and musculoskeletal diseases (RMDs). The project, known as RMD-Health, seeks to transform the way these conditions are identified and managed, offering a pathway to quicker diagnoses and more effective treatment options for individuals living with these conditions.
The initial pilot of RMD-Health is scheduled for 2026-2027, with trials planned at both the Royal Berkshire NHS Foundation Trust and Oxford University Hospitals NHS Foundation Trust. This pilot phase is critical for refining the AI system and moving it toward regulatory approval and subsequent commercialisation, ensuring that it meets the stringent requirements necessary for integration into the broader healthcare system.
The funding for this initiative is provided through a collaboration between the National Institute for Health and Care Research (NIHR), the Engineering and Physical Sciences Research Council, the Health Innovation Partnership, and Henley Business School. This financial support underscores the importance of addressing the pressing challenges associated with RMDs.
RMDs, which encompass conditions like inflammatory arthritis, affect up to one-third of the UK population and represent a leading cause of disability. These conditions place a considerable burden on both individuals and the healthcare system, often resulting in chronic pain, mobility issues, and substantial impacts on daily life.
Professor Weizi (Vicky) Li, who leads the project and serves as a professor of informatics and digital health at Henley Business School, highlighted the economic and healthcare challenges posed by RMDs. She noted, “With an estimated annual cost of £1.8 billion in sick leave and work-related disability for rheumatoid arthritis alone, the current RMD referral system faces huge challenges.”
Professor Li explained how RMD-Health aims to revolutionise the referral process, saying, “Our machine-learning system presents a new approach to RMD referrals. Unlike existing solutions, which often rely on the advice and guidance from already stretched rheumatology specialists, we’re introducing a machine learning-based decision support system enabling doctors to refer patients more accurately and promptly, ultimately leading to quicker and more effective treatment.”
One of the major issues with the current referral process is the delay in accessing specialised care for RMDs, which often results in individuals having to attend multiple GP appointments before receiving the appropriate care. Between 2019 and 2021, GPs accurately identified early inflammatory arthritis in only 40% of cases, leading to an increased workload for secondary care clinicians who must review a large volume of unnecessary referrals.
Dr. Antoni Chan, the project’s co-lead and a consultant rheumatologist at Royal Berkshire NHS Foundation Trust, emphasised the potential impact of the RMD-Health system. He stated, “This exciting and innovative project represents a major step forward in the early detection and referral of RMD, promising improved patient outcomes, reduced healthcare costs and increased efficiency across our healthcare system.” Dr. Chan also noted that during experimental trials at the trust, the AI tool demonstrated “significantly higher accuracy” than traditional clinical criteria and clinicians’ assessments.
Looking ahead, Dr. Chan expressed optimism about the project’s timeline, stating, “With this grant, we fully expect to be on track for regulatory approval at the end of three years.” The goal is to ensure that the system is ready for widespread use, offering tangible improvements in the early detection and management of RMDs.
The development of RMD-Health involves a collaborative effort that brings together AI experts, secondary care specialists, GPs, industry stakeholders, and patient representatives. This partnership aims to create a comprehensive software prototype that will lay the foundation for the future integration of RMD-Health into the NHS, ensuring that the system is both effective and practical for everyday use in clinical settings.
The project is led by Henley Business School, part of the University of Reading, in partnership with several key organisations, including the Royal Berkshire NHS Foundation Trust, the RBFT Health Data Institute, Oxford University Hospitals NHS Foundation Trust, Health Innovation Oxford and Thames Valley, Buckinghamshire, Oxfordshire and Berkshire West Integrated Care Board, and patient leaders. This consortium aims to ensure that the needs of patients remain at the forefront of the system’s development, making it as user-centred as possible.
In a related development, Flok Health, an AI-driven physiotherapy clinic tailored for people experiencing musculoskeletal issues, is set to be implemented within the NHS by autumn 2024. This initiative, announced in June 2024, aims to address the backlog in physiotherapy services and reduce waiting times for those seeking treatment. The deployment of Flok Health, alongside projects like RMD-Health, highlights the growing role of AI in transforming the delivery of healthcare for musculoskeletal conditions across the UK.
With the backing of this £1.2 million grant, the RMD-Health project is poised to make a significant difference in the early detection and referral process for RMDs, ultimately improving the quality of life for those affected and alleviating the pressure on an already strained healthcare system.
Read More
Development of new AI algorithm could revolutionise drug discovery say researchers
Researchers from King’s College London and Imperial College London have developed an innovative AI algorithm that has the potential to significantly accelerate drug discovery. This computer-based tool, known as Drug Synthesis using Monte Carlo (DrugSynthMC), is designed to enhance the diversity of chemical structures within drug libraries, creating thousands of drug-like molecules in just seconds.
Revolutionising Drug Discovery with AI
DrugSynthMC aims to overcome one of the major challenges in drug discovery: the limitations of existing drug libraries. These libraries often contain compounds that have been extensively studied and catalogued, leaving little room for the exploration of novel molecules. The new algorithm addresses this issue by generating a vast array of chemically diverse compounds, opening up new possibilities for drug candidates that may have previously been overlooked.
Dr Filippo Prischi, senior lecturer in molecular biochemistry at King’s College London and co-senior author of the study, highlighted the breakthrough:
“We showed that DrugSynthMC can expand the chemical diversity of compounds in available libraries, overcoming the limitations of existing drug collections.”
This capability is crucial because virtual-library screening—a process that relies on computational tools to sift through vast databases of known compounds—is a vital step in early-stage drug discovery. The objective is to identify chemical structures that have a high probability of binding to specific drug targets. Once promising compounds are identified, they undergo optimisation and testing in laboratory settings, both in cell cultures and animal models, before advancing to clinical trials.
Breaking Free from Traditional Limitations
Traditional methods of virtual-library screening are confined to compounds already known and catalogued. This limitation restricts researchers from discovering truly novel chemical entities, which could offer fresh approaches to treating diseases. DrugSynthMC breaks free from these constraints, allowing for the generation of new, previously unconsidered molecules.
At its core, the AI algorithm utilises a technique called Monte Carlo Tree Search, a mathematical method that calculates possible outcomes based on predefined actions. This sophisticated approach allows the algorithm to systematically build chemical structures in a simple text format. It follows a set of instructions designed to maximise the key properties of orally available drugs, such as solubility, synthesis feasibility, and safety.
Success in Creating Drug-Like Molecules
One of the remarkable achievements of DrugSynthMC is its ability to produce a large proportion of molecules that meet the desired criteria for drug development. The generated compounds are not only easy to synthesise but also soluble and non-toxic—key features for developing orally administered drugs.
The team behind this AI innovation believes that DrugSynthMC can be used to identify and refine molecules targeting proteins associated with various diseases. This could potentially lead to the discovery of novel treatments for conditions that currently have limited therapeutic options.
Their findings were published in the Journal of Chemical Information and Modeling on 9 September 2024.
A Promising Future for AI-Driven Drug Discovery
Dr Olivier Pardo, reader in cancer cell signalling at Imperial College London and co-senior author of the study, expressed his enthusiasm about the potential applications of DrugSynthMC:
“Even though this is a fairly simple algorithm, it’s far more efficient than anything more complex that has been tested or published out there and will become very useful in AI-driven drug discovery for bespoke therapeutic targets.”
One of the most promising aspects of this new tool is that it is publicly available, allowing the wider scientific community to use and build upon it. This collaborative approach could hasten progress in drug discovery, particularly in the development of treatments for diseases where current options are inadequate.
As the use of AI in drug discovery continues to grow, tools like DrugSynthMC may pave the way for faster, more efficient research, potentially leading to breakthroughs in therapeutic development for a wide range of conditions. The accessibility of this tool ensures that researchers across the globe can contribute to and benefit from this technology, propelling the field of drug discovery into a new era.
Read More
Digital health investments hold the key to preventing millions of deaths from noncommunicable diseases
An additional investment of just US$0.24 per patient per year in digital health interventions, such as telemedicine, mobile messaging, and chatbots, could save over 2 million lives from noncommunicable diseases (NCDs) over the next decade. This is according to a new report released by the World Health Organization (WHO) and the International Telecommunication Union (ITU). The report also highlights that this small investment could avert around 7 million acute medical events and hospitalisations, substantially reducing the pressure on healthcare systems globally.
The publication, titled “Going Digital for Noncommunicable Diseases: The Case for Action“, was launched during an event hosted by the Government of The Gambia at the 79th United Nations General Assembly. The launch was a collaborative effort between the ITU and WHO.
“The future of health is digital. But to make this vision a reality, we need both resources and collaboration,” stated WHO Director-General Dr Tedros Adhanom Ghebreyesus. “No single organisation can do it alone. We call on governments, partners, and donors to come together, invest strategically, and ensure that these life-saving innovations reach those who need them most.”
ITU Secretary-General Doreen Bogdan-Martin echoed this sentiment, saying, “The digital revolution has the potential to unleash a health revolution. At ITU, universal meaningful connectivity is a priority because digital is a catalyst for delivering targets in key sectors such as health and education. We call for greater collaboration between the health and tech sectors, including the development of strong digital public infrastructure, which is essential for delivering digital health services that can benefit people everywhere without leaving anyone behind.”
The Burden of Noncommunicable Diseases
Noncommunicable diseases, such as cardiovascular disease, cancer, diabetes, and chronic respiratory conditions, account for over 74% of global deaths each year. Many of these deaths are preventable. Despite progress in tackling these conditions, integrating digital health technologies into mainstream healthcare systems remains a significant challenge. The report underlines the urgency of adopting these technologies to scale up effective interventions and lessen the growing burden on healthcare systems worldwide.
Four major risk factors related to lifestyle and environment — tobacco use, unhealthy diet, harmful alcohol consumption, and physical inactivity — drive physiological changes in the body that increase the likelihood of NCDs. These factors contribute to raised blood pressure, obesity, elevated blood glucose levels, and high cholesterol. Digital tools, such as mobile messaging and chatbots, can help individuals understand and manage these modifiable risk factors, encouraging them to develop healthier habits and reduce their risk of NCDs.
The Role of Digital Health Tools
For individuals living with noncommunicable diseases, ongoing monitoring and long-term specialised care are often essential. Digital health interventions like telemedicine can help individuals overcome barriers to accessing healthcare services, such as distance or limited availability of specialists. These tools also provide real-time data for healthcare professionals, enabling them to make more informed decisions regarding their patients’ care.
While over 60% of countries have developed national digital health strategies, many face difficulties integrating these new technologies into their existing healthcare infrastructure. The report urges countries to invest in digital public infrastructure and promote the use of interoperable standards. This approach would help overcome significant obstacles to unlocking the full potential of digital health.
A Call for Global Collaboration
The report serves as a blueprint for action and supports both the WHO Global Initiative on Digital Health and the Global Strategy on Digital Health 2020-2025. The WHO, the ITU, and the United Nations Inter-Agency Task Force on the Prevention and Control of NCDs (UNIATF) Secretariat, in collaboration with the Be He@lthy, Be Mobile programme, are committed to providing tailored strategic planning and advocacy support to governments.
Dr Tedros and Doreen Bogdan-Martin emphasised the importance of collaboration between governments, tech innovators, and healthcare providers. Digital health technologies hold the promise of transforming healthcare and ensuring that individuals everywhere, regardless of location, can access the care they need. However, for this promise to be realised, both financial investment and concerted global cooperation are required.
By making digital health a central part of healthcare infrastructure, we have the potential to not only save millions of lives but also to significantly alleviate the strain on healthcare systems, particularly in low- and middle-income countries. The time for action is now. By investing strategically in digital health, we can prevent millions of unnecessary deaths and ensure a healthier future for all.
A Future Built on Digital Innovation
This report calls on governments and stakeholders to prioritise the development of digital infrastructure that supports healthcare delivery and public health initiatives. The blueprint laid out in the report highlights the need for strong, interoperable systems that can scale up interventions, ultimately leading to better health outcomes for individuals living with NCDs.
As digital technologies continue to evolve, they offer unprecedented opportunities to improve healthcare accessibility, efficiency, and effectiveness. By leveraging these innovations, we can address some of the most pressing health challenges of our time and move closer to a future where no one is left behind in the pursuit of health equity.
Read More
One in five GPs already incorporate AI tools into clinical practice, reveals BMJ survey
A recent online survey conducted by the BMJ has disclosed that 20% of UK general practitioners (GPs) are incorporating generative artificial intelligence (AI) tools, such as ChatGPT, into their clinical practice, despite the widespread lack of formal training on how to properly utilise these technologies.
The results, which were published on 17 September 2024, highlight that out of 1,006 GP respondents, 205 reported using generative AI tools in their work. Of those practitioners, 47 (29%) revealed that they utilised AI to create clinical documentation following patient consultations, while 45 (28%) used AI to suggest potential differential diagnoses.
According to the survey, ChatGPT is the most popular large language model (LLM)-based chatbot currently being used among UK GPs.
Dr Charlotte Blease, an associate professor at Uppsala University in Sweden and the study’s lead author, suggests that GPs could be finding value in these tools, particularly for assisting with administrative burdens and supporting clinical reasoning. She explained, “GPs may derive value from these tools, particularly with administrative tasks and to support clinical reasoning.”
However, Dr Blease also stressed the limitations of AI, noting that such tools can be prone to subtle errors and biases that could impact clinical decision-making. “These tools have limitations since they can embed subtle errors and biases,” she stated.
The study raised concerns over the lack of official guidance or policies concerning the use of AI in clinical practice. “Despite a lack of guidance about these tools and unclear work policies, GPs report using them to assist with their job,” Blease observed.
She further argued that the medical community needs to strike a balance by both educating current and future practitioners about the benefits of AI—such as summarising complex information—and highlighting the risks, including hallucinations, biases, and the potential compromise of patient privacy.
One of the most pressing risks she emphasised is the potential breach of patient confidentiality. “Our doctors may unintentionally be gifting patients’ highly sensitive information to these tech companies,” Dr Blease warned, underscoring the uncertainty around how companies behind generative AI models may use the data they collect.
The BMJ survey was conducted in February 2024 and distributed via Doctors.net.uk, a clinician marketing service, to a non-probability sample of GPs. This monthly ‘omnibus survey’ requires responses from 1,000 participants, all of whom are asked to complete closed-ended questions.
While GPs’ use of AI is becoming more common, another study commissioned by the Health Foundation, published in July 2024, showed broad support for AI in healthcare among both NHS staff and the general public. According to the Health Foundation’s research, 76% of NHS staff and 54% of the public are in favour of using AI for patient care.
Despite the enthusiasm, NHS England’s director of AI, imaging, and deployment, Dom Cushnan, has urged for a cautious and evidence-based approach. Speaking at the Digital Health AI and Data event in October 2023, Cushnan underscored the need for health systems to ensure that AI tools are suitable and rigorously tested before being fully integrated into clinical settings.
While Cushnan acknowledged the excitement surrounding AI’s potential, he also emphasised that such technology must pass stringent reviews to ensure its efficacy and safety for patient care. “AI tools must undergo a rigorous process before they can be integrated into clinical practice,” Cushnan said, reinforcing the importance of evidence-based approaches in healthcare innovations.
This growing integration of AI into the medical profession highlights a significant shift in healthcare, yet it also raises critical questions about training, patient safety, and ethical considerations as the technology continues to evolve.
Read More
NHS Providers urges government to prioritise digital investment to boost productivity
NHS Providers has called for urgent government investment in digital technology as a means to enhance productivity, following the release of a survey involving leaders of NHS trusts. This call comes amid growing concerns about financial pressures and operational challenges facing the health service.
The survey, whose results were published on 8 September 2024, painted a stark picture of the financial health of the NHS. Trust leaders indicated that their finances have been severely impacted by a number of factors, including ongoing industrial action, the effects of inflation, demands for increased efficiency, and the overall operational strain on the NHS. These challenges have exacerbated existing financial pressures, placing trust leaders in a difficult position as they attempt to manage their budgets for the upcoming year.
The survey found that a significant 51% of respondents were “extremely concerned” about their ability to meet operational priorities within their respective organisation’s financial budget for the 2024/25 fiscal year. This concern highlights the precarious financial state of many NHS trusts as they face increasing demand for services with limited resources.
Moreover, 92% of those surveyed expressed the view that the efficiency challenge for 2024/25 is more daunting than in the previous year, with the need to do more with less becoming ever more critical. This sentiment reflects the growing pressure on NHS leaders to find ways to increase efficiency in an already overstretched system.
When it comes to meeting recovery targets, trust leaders were notably divided in their confidence levels. While almost a third of respondents (32%) were optimistic that their system would achieve its targets to reduce waiting times for physical health services in 2024/25, only 8% felt confident that similar targets for mental health services would be met. This disparity underscores the particular difficulties the NHS faces in addressing long waits in mental health care, a sector that continues to experience significant challenges.
Respondents also shared their views on accident and emergency (A&E) services, with 46% of those providing these services indicating that their trust is likely to meet the new target of seeing 78% of A&E attendees within four hours by March 2025. This reflects the ongoing challenges in delivering timely emergency care, as the NHS grapples with rising demand and workforce shortages.
On the financial front, the outlook is similarly divided. A significant proportion of respondents (44%) forecasted a deficit for their trust’s financial position in 2024/25, while 45% predicted a breakeven position, and only 11% anticipated a surplus. These figures highlight the fragile financial footing on which many NHS trusts currently stand.
Sir Julian Hartley, Chief Executive of NHS Providers, praised the resilience and adaptability of trust leaders and their teams in the face of these considerable challenges. He stated: “It’s incredibly impressive how trust leaders and their teams are responding to the productivity challenge. You can see the benefits of collaboration, incentivising staff, and using digital tools to help them work more efficiently.” However, Sir Julian emphasised that these efforts, while commendable, can only achieve so much without substantial strategic investment.
He continued: “That’s all happening, but it can only go so far without significant strategic investment in infrastructure and digital technologies. We need to recognise the value of investment in the NHS – its impact as a catalyst for economic growth, employment, and research, and the importance of funding infrastructure, digital, and other technology to drive productivity.”
The timing of these survey findings is significant, coming just ahead of a much-anticipated review of the NHS by esteemed NHS surgeon and independent peer, Lord Ara Darzi. Commissioned by Health Secretary Wes Streeting, this review is expected to be published on 12 September 2024. Early indications suggest that the report will include sharp criticism of the NHS’s productivity levels, as well as concerns about the state of children’s health.
Prime Minister Sir Keir Starmer also weighed in on the state of the NHS during an interview broadcast by the BBC on 8 September 2024. In his remarks, the Prime Minister described the NHS as having been “broken” by successive Conservative governments. He referenced the upcoming review, underscoring the severity of the challenges facing the NHS.
Responding to the Prime Minister’s comments, Sir Hartley expressed that these concerns mirrored those frequently raised by NHS trust leaders. He said: “The Prime Minister’s comments today echo what trust leaders have been telling us. The shockwaves of the longest and deepest squeeze in NHS financial history, a growing mismatch between capacity and demand, major workforce challenges, and the after-effects of the Covid-19 pandemic are still being felt throughout the health service.”
Sir Hartley went on to highlight the need for sustained, long-term financial investment in the NHS, pointing out that the current approach to funding is unsustainable. He stated: “Time and again, trust leaders tell us they want to see long-term, multi-year investment in the health service which allows them to plan for the future instead of this stop-start approach to NHS funding which leaves them constantly worrying about budget cuts followed by quick-fix, short-term funding announcements.”
Adding further weight to the conversation around NHS productivity, a report published by the Health Foundation in April 2024 examined clinicians’ views on the technologies that could offer the greatest opportunities to enhance productivity within the NHS. The report underscored the potential of digital tools to drive efficiencies, streamline operations, and improve patient outcomes, aligning closely with the calls from NHS Providers for greater investment in this area.
In conclusion, the message from NHS Providers is clear: while trust leaders and their teams are doing all they can to meet the challenges they face, there is a pressing need for the government to step up with significant investment in digital technologies and infrastructure. Such investment is critical not only for improving productivity but also for securing the long-term future of the NHS as it continues to navigate unprecedented operational and financial pressures.
Read More
AI-powered stethoscope doubles success in diagnosing heart failure during pregnancy
Heart failure during pregnancy is a life-threatening and often overlooked condition, primarily because its symptoms—such as shortness of breath, severe fatigue, and difficulty breathing while lying down—closely resemble typical discomforts associated with pregnancy. This confusion often leads to delayed diagnosis. However, a groundbreaking study presented at the European Society of Cardiology Congress, based on research by the Mayo Clinic, demonstrates that an artificial intelligence (AI)-enabled digital stethoscope allowed healthcare providers to diagnose twice as many cases of heart failure compared to conventional obstetric care methods. The full study has been published in Nature Medicine.
This clinical trial was conducted in Nigeria, where pregnancy-related heart failure occurs more frequently than in any other region worldwide. The findings revealed that the AI-enabled stethoscope was 12 times more likely to detect weakened heart function—specifically in cases with an ejection fraction of less than 45%—compared to traditional methods. An ejection fraction under 45% is a critical indicator of peripartum cardiomyopathy, a form of heart failure that can develop during the final months of pregnancy or soon after childbirth.
“Early detection of this form of heart failure is crucial for safeguarding maternal health and wellbeing,” explained Dr Demilade Adedinsewo, a cardiologist at the Mayo Clinic and the lead investigator of the study. “Symptoms of peripartum cardiomyopathy can progressively worsen as pregnancy advances, or more commonly after childbirth. If left undiagnosed and untreated, this condition can become life-threatening as the heart weakens further. Although medications can help when detected early, severe cases may necessitate advanced interventions, including intensive care, mechanical heart pumps, or even heart transplants in extreme situations.”
The randomised, controlled, open-label clinical trial involved nearly 1,200 participants. Each was screened for heart conditions using either standard obstetric care or AI-enhanced tools. Researchers at the Mayo Clinic had previously developed a 12-lead AI-electrocardiogram (ECG) algorithm, capable of predicting a weak heart pump, known clinically as low ejection fraction. This algorithm was further refined by Eko Health, which incorporated it into its point-of-care digital stethoscope. The stethoscope, cleared by the U.S. Food and Drug Administration (FDA), is designed to detect heart failure in patients with low ejection fractions.
The results of the study were compelling. The combination of the AI-based screening tools—comprising the digital stethoscope and the 12-lead ECG—enabled doctors to identify cases of weak heart function with a high degree of accuracy. Specifically, the AI-enhanced stethoscope doubled the number of heart failure cases detected with ejection fractions below 50%, and significantly increased detection rates for ejection fractions under 45%.
The researchers evaluated the AI-enabled screening tools across three different levels of ejection fraction, all of which are used in the clinical diagnosis of heart failure. An ejection fraction below 45% is the threshold for diagnosing peripartum cardiomyopathy, while a measurement below 40% indicates heart failure with reduced ejection fraction, for which specific medications are known to alleviate symptoms and lower the risk of mortality. Ejection fractions below 35% suggest critically low heart pump function, often requiring aggressive management, including advanced heart failure treatments or the implantation of a defibrillator if heart function fails to improve. Each participant in the intervention group underwent an echocardiogram at the start of the trial, which provided confirmation of the AI-predicted heart function.
“This research provides compelling evidence that AI-assisted tools can significantly improve the detection of peripartum cardiomyopathy, especially in Nigerian women, where the condition is more prevalent,” stated Dr Adedinsewo. “However, there are still important questions that need to be addressed. Our next step involves assessing the usability and adoption of these tools by Nigerian healthcare providers, including both doctors and nurses, as well as evaluating the impact of the AI-enabled stethoscope on patient outcomes. In the United States, peripartum cardiomyopathy affects approximately 1 in 2,000 women, but among African American women, the incidence is as high as 1 in 700. Evaluating the effectiveness of this AI tool in the U.S. will further test its capabilities across diverse populations and healthcare environments.”
The clinical trial received financial backing from several sources, including the Mayo Clinic’s Centres for Digital Health and Community Health and Engagement Research, the Mayo Clinic’s Building Interdisciplinary Research Careers in Women’s Health (BIRCWH) programme, which is funded by the National Institutes of Health (NIH), and the Mayo Clinic’s Centre for Clinical and Translational Sciences (CCATS), also funded by the NIH.
The study not only underscores the potential of AI in improving maternal healthcare, but also highlights the critical importance of early diagnosis in preventing life-threatening complications related to heart failure during and after pregnancy. With further refinement and wider implementation, this AI-enhanced tool could transform the way pregnancy-related heart failure is detected and managed, potentially saving countless lives in the process.
Read More
Digital consultations significantly enhance medication optimisation in heart failure patients
Heart failure, a condition impacting over 60 million people globally, presents a significant challenge in ensuring patients receive the most effective combination of medications. A recent study has revealed that patients are four times more likely to achieve optimal medication regimens after just 12 weeks of engaging in digital consultations. This groundbreaking research, led by a collaboration of five Dutch hospitals under the coordination of Amsterdam UMC, demonstrates that digital consultations not only improve the quality of care but also maintain high levels of patient satisfaction. These findings have been published in Nature Medicine and were also presented at the European Society of Cardiology’s annual conference.
Reflecting on the origins of the study, Mark Schuuring, a former cardiologist at Amsterdam UMC and currently practising at Medical Spectrum Twente, stated, “During the COVID-19 pandemic, almost all of our patients were suddenly digital consult patients and, to be honest, this worked well but there were also concerns. Those concerns gave us the idea for this study.”
The programme developed by the researchers measures the quality of care by comparing the treatment approaches taken during digital consultations with the most current medical guidelines. Schuuring elaborated, “We investigated the digital data exchange between patients and doctors and provided them both with more information. The programme encourages doctors and nurses to give treatment that is closest to international guidelines. The business community makes extensive use of such programmes, but they are not yet commonplace in the care sector.”
The study involved 150 heart failure patients who were randomly assigned to one of two groups: one group participated in a digital consultation strategy, while the other received traditional care. After 12 weeks, the researchers assessed how many patients in each group had achieved the optimal combination of medications. The results were striking: 28% of patients in the digital consultation group reached optimal medication levels, compared to only 7% in the traditional care group.
Schuuring emphasised the significance of these findings, stating, “Ultimately, we saw that this is superior to the way we currently organise care and this is demonstrated by the data.”
In addition to evaluating the effectiveness of digital consultations in improving medication optimisation, the researchers also addressed common concerns associated with digital healthcare. They found no significant differences in the time invested by healthcare providers, patient satisfaction levels, or, most importantly, the patients’ quality of life.
“This study shows that digital consultations are really a win-win; the patient’s care was improved, and their experience was also not in any way reduced,” Schuuring concluded. “We think that this could work way beyond heart failure. Something which is urgently needed as the increases in patient populations outstrip the growth in staff numbers.”
This study highlights the potential for digital consultations to play a crucial role in the future of healthcare, particularly in managing chronic conditions such as heart failure. The findings suggest that with the right digital tools, healthcare providers can offer high-quality care that adheres closely to international guidelines, ultimately improving patient outcomes while also addressing the growing demands on healthcare systems worldwide.
Read More